No products in the cart.
Even though LASIK has been around for 20 years in the U.S., one former FDA adviser has said the procedure should be taken off the market.
Dr. Morris Waxler originally voted to approve the eye surgery, but he’s changed his mind.
“Essentially we ignored the data on vision distortions that persisted for years,” the retired FDA adviser told CBS News.
“I re-examined the documentation … and I said, ‘Wow this is not good,'” he added.
AMERICANS HAVE SPENT BILLIONS TREATING DIABETES
LASIK surgery uses a special laser to reshape the cornea to improve the eye’s “focusing power,” according to the U.S. Food and Drug Administration (FDA).
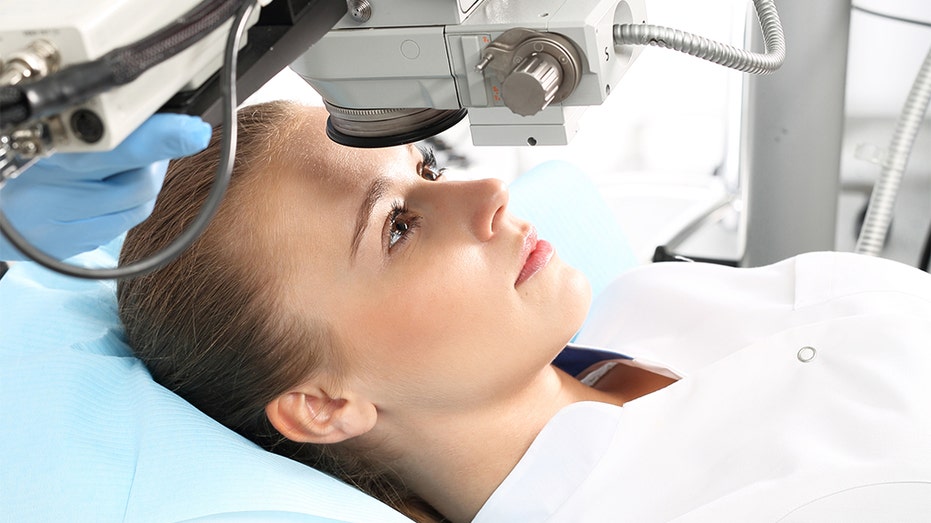
The agency conducted a survey through its LASIK Quality of Life Collaboration Project and found that 95 percent of participants were satisfied with their vision after the procedure, the FDA says on its website.
US SUPERBUG INFECTIONS ON THE RISE, BUT DEATHS ARE FALLING
However, Waxler told CBS that he believes complications rates for the procedure are between 10 and 30 percent.
On its website, the FDA does explain many of the symptoms that participants experience after LASIK, including halos, dry eyes, starbursts, ghosting and glare.
It also explains the risks of LASIK surgery, which include loss of vision, debilitating visual symptoms that seriously affect vision at night and severe dry eye syndrome.
GET FOX BUSINESS ON THE GO BY CLICKING HERE
The FDA also says that because the first laser was only approved in 1998, “the long-term safety and effectiveness of LASIK surgery is not known.”
Back in 2011, Waxler petitioned the FDA to recall LASIK, but the agency denied the petition, CBS reported.
Though the agency did not immediately respond to FOX Business’ request for comment Thursday night, it told CBS that it hasn’t “found any new safety concerns associated with LASIK devices.”
CLICK HERE TO READ MORE ON FOX BUSINESS
“There’s nothing wrong with a person’s eyes who goes to get LASIK,” Waxler told the television station. “They have healthy eyes. They could go and get a pair of glasses.”


